Neurocrine Biosciences’ lightning-fast $2.9 billion takeover of Soleno Therapeutics (NASDAQ: SLNO) isn’t your typical biotech M&A deal. It’s a full-blown repricing event that just lit a fire under an entirely new corner of the obesity market.
The prize? Prader-Willi syndrome (PWS) – a rare, brutal neuroendocrine disorder marked by uncontrollable hunger and severe obesity. Soleno’s Vykat XR, approved last year, racked up roughly $190 million in its first full year on the market. That alone justified the price tag. But the real message is bigger: a big-cap biotech just dropped nearly $3 billion on a non-GLP-1 obesity play. Rare, brain-driven forms of obesity are no longer a sideshow – they’re a high-value category Wall Street can’t ignore anymore.
The hunt for the next wave of companies with exposure to this category is already underway – yet public companies with direct exposure remain surprisingly scarce. Palatin Technologies (NYSE: PTN) seems to be sitting right in the sweet spot.
MC4R Is Proven – But First-Gen Drugs Have Real Problems
Palatin is all-in on the melanocortin-4 receptor (MC4R) – the brain’s master switch for appetite and satiety. This isn’t early-stage guesswork.
Rhythm Pharmaceuticals has already turned the pathway into a $194 million (and growing) revenue machine. FDA decisions have now green-lit MC4R drugs in hypothalamic obesity, locking in the biology as commercially viable.
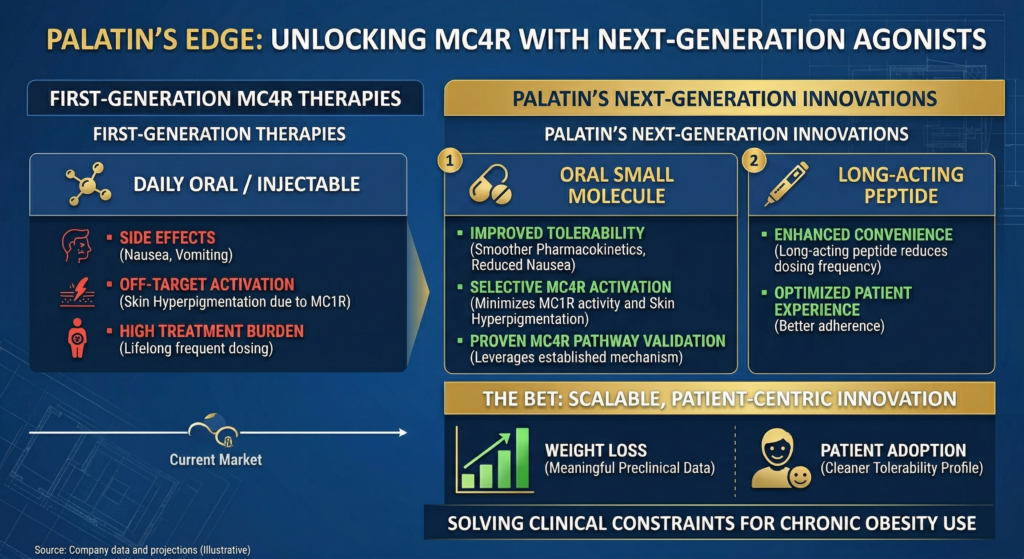
The catch? The first wave of MC4R therapies exposed clear limitations that hurt long-term use: nausea and vomiting tied to peak drug concentrations, skin hyperpigmentation from off-target MC1R activation (seen in a majority of patients in some trials), and daily injections that become a drag for lifelong therapy.
Even Soleno’s approved PWS drug requires monitoring for hyperglycemia and fluid retention. These medicines work – but they’re not yet ideal for chronic, real-world adoption.
Palatin’s Edge: Next-Gen MC4R Agonists Built to Fix the Flaws
Palatin isn’t chasing discovery breakthroughs. It’s laser-focused on solving the exact pain points that have held the category back.
The Company is advancing two differentiated MC4R agonists: a once-daily oral small molecule designed for strong MC4R activation with reduced MC1R activity and smoother pharmacokinetics to cut nausea and hyperpigmentation, plus a long-acting weekly injectable peptide for even better convenience.
Preclinical data on its oral small molecule showed meaningful MC4R-driven weight loss with a cleaner tolerability profile. Early, yes – but the program was engineered around known clinical constraints, not theoretical ones.
In biotech, the biggest winners are often the ones that take a validated mechanism and make it usable at scale. That’s Palatin’s bet.
A Market That’s Suddenly Worth Fighting For
Neurocrine’s move doesn’t just validate PWS – it proves rare neuroendocrine obesity disorders are commercially attractive. These are identifiable patient populations managed by specialists and requiring chronic, lifelong treatment – the perfect recipe for pricing power, strong reimbursement, and durable revenue that strategic buyers crave.
Palatin has positioned its pipeline squarely in the bullseye, with development plans now covering both hypothalamic obesity (lead indication) and Prader-Willi syndrome. The treatment landscape is wide open for improvement, and competition is shifting toward better tolerability and dosing.
The Valuation Gap Is Getting Hard to Ignore
Stack it up: Rhythm’s MC4R franchise commands a multi-billion-dollar valuation, while Neurocrine just paid $2.9 billion for one approved PWS asset.
Palatin, with two next-generation MC4R agonist candidates heading into the clinic in 2026, trades at a tiny fraction of those benchmarks.
That’s not an accident – it’s stage and timing. IND filings are expected later this year, with first human data to follow. In a freshly validated category, the move from preclinical to clinical is often when the sharpest re-ratings happen.
Risks are real. Biotech at these stages is volatile, and preclinical promise doesn’t guarantee success. But the macro setup has flipped. The $2.9 billion deal answered the “is this a real market?” question – decisively.
Now the only question left is: who builds the better mousetrap? Palatin is already there – at a valuation that that seems to still allow for major upside.
Investors looking for asymmetric exposure in the evolving obesity landscape may want to keep PTN high on the watchlist. The next wave in rare, brain-driven obesity is just getting started – and Palatin seems to be built for exactly what comes next.
Read this Next >> Advanced Biomed to Acquire Acellent Technologies (Hong Kong) for $1.08 Million in Stock (ADVB)
Recent News Highlights from Palatin (PTN):
Palatin Reports Second Quarter Fiscal Year 2026 Financial Results and Provides Corporate Update
Palatin Reports First Quarter Fiscal Year 2026 Financial Results and Provides Corporate Update
Important Disclaimers and Disclosures: The author, Wall Street Wire, is a content and media technology platform that connects the market with under-the-radar companies. The platform operates a network of industry-focused media channels spanning finance, biopharma, cyber, AI, and additional sectors, delivering insights on both broader market developments and emerging or overlooked companies. Wall Street Wire is not a broker-dealer or investment adviser. References to market size estimates, price targets, or other third-party data are provided strictly for informational purposes. Wall Street Wire receives cash compensation from Palatin Technologies Inc for coverage and awareness services, which are provided on an ongoing subscription basis. The content above is a form of paid advertising and promotion and is for informational purposes only and does not constitute financial or investment advice. Full compensation details, information about the operator of Wall Street Wire, and the complete set of disclaimers and disclosures applicable to this content are available at: wallstwire.ai/disclosures. This article should not be considered an official communication of the issuer.
